This N0 molecule, right? Which is given to us. Before we turn our attention to the MO diagram of dioxygen. You can just try to connect these orbital's with each other and we are going to get a structure, something like this. A fundamental rule of molecular orbital theory is that the number of molecular orbitals must. This is the correct view of forming this bonding forming this molecule little diagram, right? You can just connect the dots here. Right? So we can just fill them in this way, we can just fill them in this way. And then we have this last week sigma star which is the anti bonding sigma bonding molecular orbital anti bonding sigma molecular it'll basically, Right, so we can just fill this portion with four electrons because there are only four electrons and in this portion there are a total of seven electrons. And then we have this five star, two P which is anti bonding pi bond, anti bonding PMO. So if we can just draw the molecular orbital, we can say we will be having this sigma, we believe this sigma to west and then we have this sigma star to west and then we have this five to P. Two and two P four is the electronic configuration for oxygen. Two and two P three is the electronic configuration for nitrogen by two S. Right? So now we can say there is two ways of nitrogen and this is the two p orbital off nitrogen, right? And similarly the oxygen atom would also be having the two S and two P orbiters but they will be situated much lower than the orbiters of nitrogen in the order of energy right now we can say to S. Two because this is not going to make any difference in the M. Right now, we can just write the balance shell potential configuration of both of these items, right? We can just ignore one S. 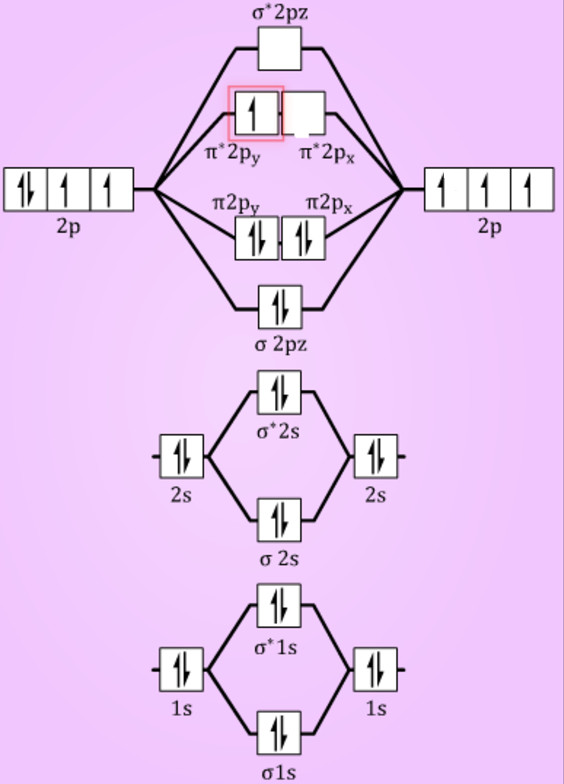
Right? So we can just say that, let us say nitrogen is shown on the left hand side and oxygen is shown on the right hand side. Right now, the important factor is that while these and oh attempts or begins and nitrogen oxygen atoms approach each other and their atomic orbital is combined with each other to get to give new molecular orbiters, we can clearly say that since the electro negativity of oxygen is better than the electron negativity value of nitrogen, the atomic capitals of the oxygen atom would be situated much below that, much lower than the atomic levels of nitrogen. So we can say that for nitrogen the atomic configuration of weekends, the electronic configuration will be one is two, US two and two P three for oxygen, it is one S 22 S. Right? Because in this molecule we can say both of these items are different. And oh right, so we can say this is hetero atomic, this is an hetero atomic ah and die nuclear hetero hetero, atomic dying nuclear molecule. In this question, we are expected to draw a molecular orbital diagram for these species.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
